
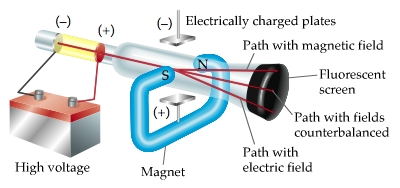
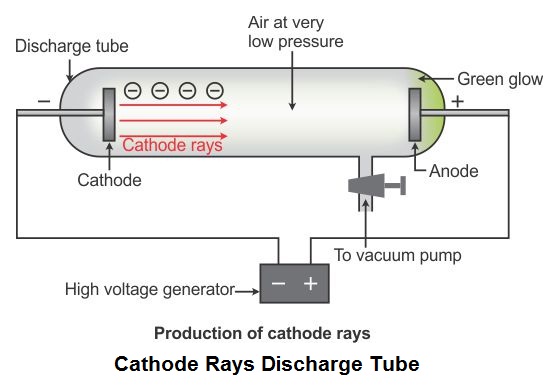
Since the electrons have a negative charge, they are repelled by the negative cathode and attracted to the positive anode. The increased random heat motion of the filament knocks electrons out of the surface of the filament, into the evacuated space of the tube. Modern vacuum tubes use thermionic emission, in which the cathode is made of a thin wire filament which is heated by a separate electric current passing through it. The positive ions were accelerated by the electric field toward the cathode, and when they collided with it they knocked electrons out of its surface these were the cathode rays. In the early experimental cold cathode vacuum tubes in which cathode rays were discovered, called Crookes tubes, this was done by using a high electrical potential of thousands of volts between the anode and the cathode to ionize the residual gas atoms in the tube. To release electrons into the tube, they first must be detached from the atoms of the cathode. The Maltese cross has no external electrical connection.Ĭathode rays are so named because they are emitted by the negative electrode, or cathode, in a vacuum tube. Cathode-ray tubes (CRTs) use a focused beam of electrons deflected by electric or magnetic fields to render an image on a screen.Ī diagram showing a Crookes tube connected to a high voltage supply.
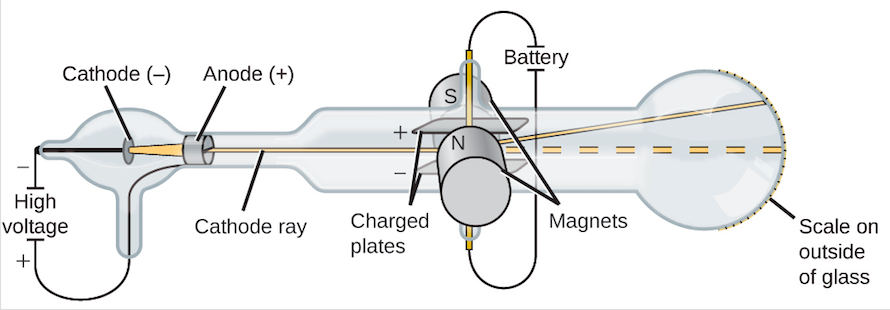
Thomson showed that cathode rays were composed of a previously unknown negatively charged particle, which was later named the electron. They were first observed in 1859 by German physicist Julius Plücker and Johann Wilhelm Hittorf, and were named in 1876 by Eugen Goldstein Kathodenstrahlen, or cathode rays. If an evacuated glass tube is equipped with two electrodes and a voltage is applied, glass behind the positive electrode is observed to glow, due to electrons emitted from the cathode (the electrode connected to the negative terminal of the voltage supply). Cathode rays are normally invisible in this demonstration with a Teltron tube, enough gas has been left in the tube that the gas atoms luminesce when struck by the fast-moving electrons.Ĭathode rays ( electron beam or e-beam) are streams of electrons observed in discharge tubes. Since he knew the tube was empty those particles had to come from the atoms of the anode, implying that there were tiny negatively charged particles inside the atoms that make up everything.A beam of cathode rays in a vacuum tube bent into a circle by a magnetic field generated by a Helmholtz coil. He measured how far the beam moved in response to a known charge and calculated the mass of the beam, and later the individual charge and mass of the particles that made it up. With a little bit of math he deduced the ray was not a wave, but a beam of particles flowing from the anode to the cathode that were negatively charged. Thompson improved the cathode ray tube so that he could deflect the beam with a known electric field and measure how far the beam was deflected. Many scientists of the day believed the cathode ray as it was called was a wave that had no mass. The beam moves around when you do this! Now they had known for a bit that electric and magnetic fields were intertwined and that a magnetic field can push electrically charged objects or particles. What Thompson did was put a magnet next to the beam. This greenish stream of light grows out from the anode and reaches toward the cathode until it connects and this green beam is formed in the middle. When you charge one wire with a positive electric charge (the anode) and the other wire a negative charge (a cathode) something really cool happens. If you take a cylinder of glass and place two wires at either end, then evacuate the gas inside so it's just a vacuum you create something called a cathode ray tube.
